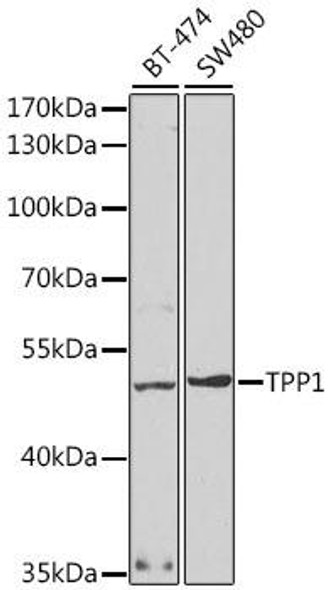
| UniProt Protein Function: | TPP1: Lysosomal serine protease with tripeptidyl-peptidase I activity. May act as a non-specific lysosomal peptidase which generates tripeptides from the breakdown products produced by lysosomal proteinases. Requires substrates with an unsubstituted N-terminus. Defects in TPP1 are the cause of neuronal ceroid lipofuscinosis type 2 (CLN2). A form of neuronal ceroid lipofuscinosis. Neuronal ceroid lipofuscinoses are progressive neurodegenerative, lysosomal storage diseases characterized by intracellular accumulation of autofluorescent liposomal material, and clinically by seizures, dementia, visual loss, and/or cerebral atrophy. The lipopigment pattern seen most often in CLN2 consists of curvilinear profiles. Belongs to the peptidase S53 family. 2 isoforms of the human protein are produced by alternative splicing. |
| UniProt Protein Details: | Protein type:Secreted; Protease; EC 3.4.14.9; Secreted, signal peptide; Mitochondrial Chromosomal Location of Human Ortholog: 11p15 Cellular Component: lysosomal lumen; mitochondrion; lysosome; melanosome Molecular Function:peptidase activity; tripeptidyl-peptidase activity; protein binding; serine-type peptidase activity; serine-type endopeptidase activity; metal ion binding; endopeptidase activity; peptide binding Biological Process: nervous system development; cellular protein metabolic process; unfolded protein response, activation of signaling protein activity; epithelial cell differentiation; unfolded protein response; lysosome organization and biogenesis; peptide catabolic process; protein catabolic process; lipid metabolic process; neuromuscular process controlling balance; proteolysis; bone resorption Disease: Ceroid Lipofuscinosis, Neuronal, 2 |
| NCBI Summary: | This gene encodes a member of the sedolisin family of serine proteases. The protease functions in the lysosome to cleave N-terminal tripeptides from substrates, and has weaker endopeptidase activity. It is synthesized as a catalytically-inactive enzyme which is activated and auto-proteolyzed upon acidification. Mutations in this gene result in late-infantile neuronal ceroid lipofuscinosis, which is associated with the failure to degrade specific neuropeptides and a subunit of ATP synthase in the lysosome. [provided by RefSeq, Jul 2008] |
| UniProt Code: | O14773 |
| NCBI GenInfo Identifier: | 108936017 |
| NCBI Gene ID: | 1200 |
| NCBI Accession: | O14773.2 |
| UniProt Secondary Accession: | O14773,Q53HT1, Q5JAK6, Q6UX56, Q71JP6, Q96C37, |
| UniProt Related Accession: | O14773 |
| Molecular Weight: | 563 |
| NCBI Full Name: | Tripeptidyl-peptidase 1 |
| NCBI Synonym Full Names: | tripeptidyl peptidase I |
| NCBI Official Symbol: | TPP1 |
| NCBI Official Synonym Symbols: | CLN2; GIG1; LPIC; SCAR7; TPP-1 |
| NCBI Protein Information: | tripeptidyl-peptidase 1; tripeptidyl aminopeptidase; growth-inhibiting protein 1; cell growth-inhibiting gene 1 protein; lysosomal pepstatin insensitive protease |
| UniProt Protein Name: | Tripeptidyl-peptidase 1 |
| UniProt Synonym Protein Names: | Cell growth-inhibiting gene 1 protein; Lysosomal pepstatin-insensitive protease; LPIC; Tripeptidyl aminopeptidase; Tripeptidyl-peptidase I; TPP-I |
| Protein Family: | Tripeptidyl-peptidase |
| UniProt Gene Name: | TPP1 |
| UniProt Entry Name: | TPP1_HUMAN |